Diagnostic Accuracy of Sonographic Signs of Extrapulmonary Tuberculosis and Treatment Response Monitoring in HIV-Positive and -Negative Populations
Keywords:
eFASH, FASH, ultrasound, extrapulmonary tuberculosis, treatment monitoringAbstract
Background
Diagnosis and monitoring of extrapulmonary tuberculosis (EPTB) remains challenging. Ultrasound such as the extended focused assessment for HIV-associated tuberculosis (eFASH) protocol might improve diagnosis and monitoring of treatment responses. This study determined the diagnostic accuracy of eFASH for EPTB and its value in monitoring EPTB treatment response compared with clinical signs and symptoms.
Methods
We performed a post-hoc analysis of a trial assessing eFASH impact on management of adults with suspected EPTB. Participants who had baseline and follow-up ultrasound examinations were included. We assessed the diagnostic accuracy of eFASH and compared the evolution of eFASH and clinical signs and symptoms in participants with definite EPTB, stratified by favorable treatment outcomes at 6 months.
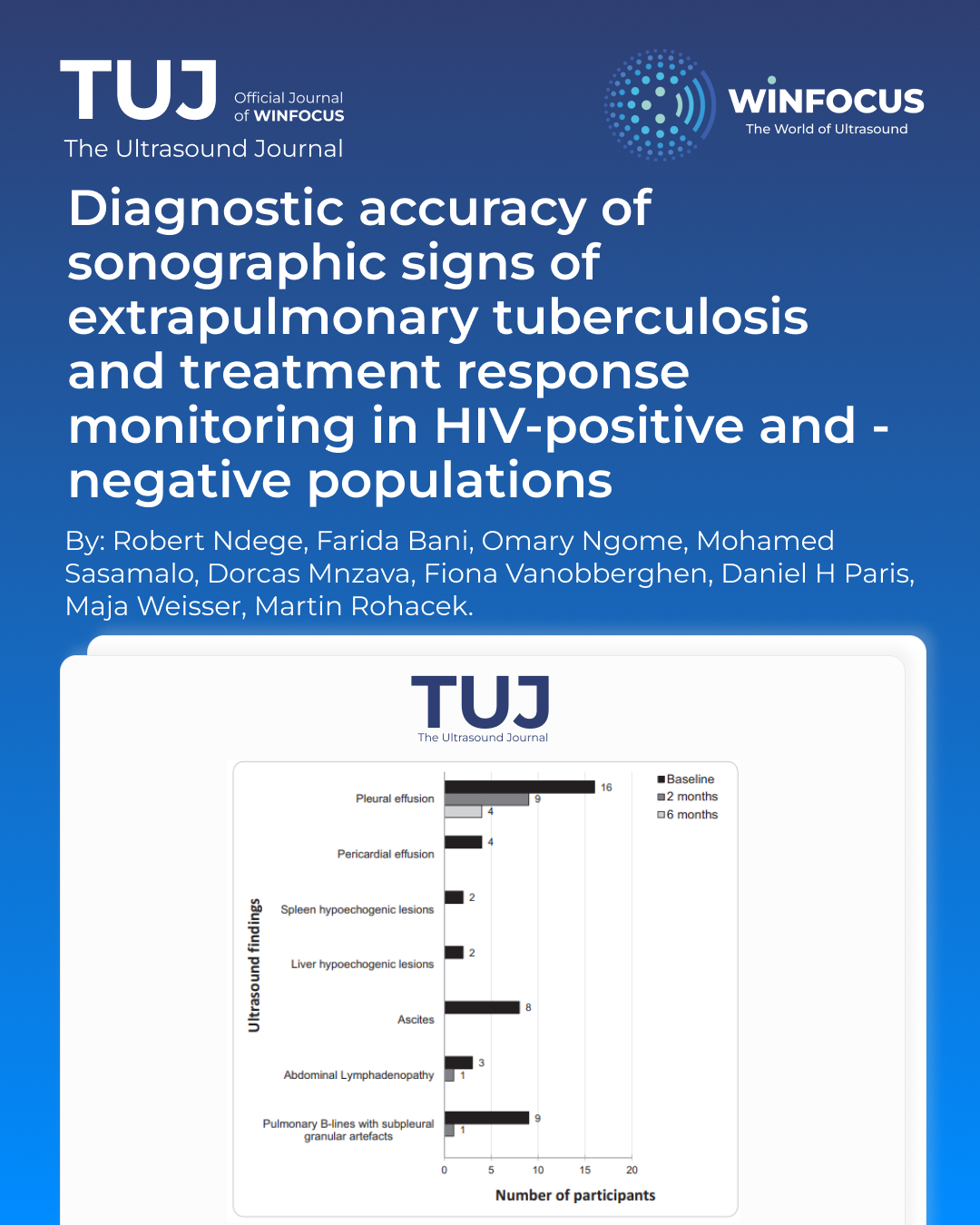
Results
In 296 included participants (95 with definite EPTB, 201 with no definite EPTB), the most common eFASH signs were pleural effusion (47%) and pulmonary B-lines with subpleural granular artefacts (34%). Pleural effusion was the only sign that persisted beyond 6 months. eFASH had a sensitivity of 93.7% (95% CI, 86.8-97.6) and a specificity of 37.8% (95% CI, 31.1- 44.9) for definite EPTB. At 2 months, favorable outcomes were similar between participants with full and partial resolution of eFASH signs (83% versus 81%). In contrast, a higher proportion of favorable outcomes was seen in participants with full resolution of clinical signs and symptoms (90% versus 60%).
Conclusion
eFASH shows high sensitivity but low specificity for definite EPTB. Ultrasound can be used alongside clinical signs and symptoms to monitor treatment response in patients with EPTB.
References
1. Global tuberculosis report 2024. Geneva: World Health Organization; 2024. Licence: CC BY-NC-SA 3.0 IGO.
2. WHO consolidated guidelines on tuberculosis. Module 5: management of tuberculosis in children and adolescents. Geneva: World Health Organization; 2022. Licence: CC BY-NC-SA 3.0 IGO.
3. McNally E, Ross C, Gleeson LE. The tuberculous pleural effusion. Breathe (Sheff) 2023;19(4):230143. DOI: 10.1183/20734735.0143-2023.
4. Zurcher K, Ballif M, Kiertiburanakul S, et al. Diagnosis and clinical outcomes of extrapulmonary tuberculosis in antiretroviral therapy programmes in low- and middle-income countries: a multicohort study. J Int AIDS Soc 2019;22(9):e25392. DOI: 10.1002/jia2.25392.
5. Ndege R, Ngome O, Vanobberghen F, et al. Ultrasononography in Managing Extrapulmonary Tuberculosis: A Randomized, Controlled, Parallel, Superiority, Open-Label Trial. Clin Infect Dis 2023;76(6):1013-1021. DOI: 10.1093/cid/ciac871.
6. Ndege R, Rohacek M, Bani F, et al. Diagnostic Yield of Urine Xpert MTB/RIF Ultra in Adults With Suspected Extrapulmonary Tuberculosis. Open Forum Infect Dis 2024;11(7):ofae338. (In eng). DOI: 10.1093/ofid/ofae338.
7. Van Hoving DJ, Griesel R, Meintjes G, Takwoingi Y, Maartens G, Ochodo EA. Abdominal ultrasound for diagnosing abdominal tuberculosis or disseminated tuberculosis with abdominal involvement in HIV-positive individuals. Cochrane Database Syst Rev 2019;9(9):Cd012777. (In eng). DOI: 10.1002/14651858.CD012777.pub2.
8. Van Hoving DJ, Kenge AP, Maartens G, Meintjes G. Point-of-Care Ultrasound Predictors for the Diagnosis of Tuberculosis in HIV-Positive Patients Presenting to an Emergency Center. J Acquir Immune Defic Syndr 2020;83(4):415-423. DOI: 10.1097/QAI.0000000000002279.
9. Theron G, Venter R, Smith L, et al. False-Positive Xpert MTB/RIF Results in Retested Patients with Previous Tuberculosis: Frequency, Profile, and Prospective Clinical Outcomes. J Clin Microbiol 2018;56(3). DOI: 10.1128/JCM.01696-17.
10. World Health O, World Health O. Treatment of tuberculosis: guidelines. 4th edition ed. Geneva: World Health Organization; 2010.
11. WHO operational handbook on tuberculosis Module 4: Treatment – drug-susceptible tuberculosis treatment. Geneva: World Health Organization; 2022. Licence: CC BY-NC-SA 3.0 IGO.
12. Heller T, Wallrauch C, Brunetti E, Giordani MT. Changes of FASH ultrasound findings in TB-HIV patients during anti-tuberculosis treatment. Int J Tuberc Lung Dis 2014;18(7):837-9. DOI: 10.5588/ijtld.13.0029.
13. Bosch-Marcet J, Serres-Creixams X, Borras-Perez V, Coll-Sibina MT, Guitet-Julia M, Coll-Rosell E. Value of sonography for follow-up of mediastinal lymphadenopathy in children with tuberculosis. J Clin Ultrasound 2007;35(3):118-24. DOI: 10.1002/jcu.20304.
14. Ndege R, Ngome O, Bani F, et al. Ultrasound in managing extrapulmonary tuberculosis: a randomized controlled two-center study. BMC Infect Dis 2020;20(1):349. DOI: 10.1186/s12879-020-05073-9.
15. Heller T, Wallrauch C, Goblirsch S, Brunetti E. Focused assessment with sonography for HIV-associated tuberculosis (FASH): a short protocol and a pictorial review. Crit Ultrasound J 2012;4(1):21. (In eng). DOI: 10.1186/2036-7902-4-21
16. McHugh ML. Interrater reliability: the kappa statistic. Biochem Med (Zagreb) 2012;22(3):276-82. (In eng).
17. WHO. Guidelines for Intensified Tuberculosis Case Finding and Isoniazid Preventive Therapy for People Living with HIV in Resource Constrained Settings. Geneva, Switzerland: World Health Organization; 2010.
18. Weber SF, Saravu K, Heller T, et al. Point-of-Care Ultrasound for Extrapulmonary Tuberculosis in India: A Prospective Cohort Study in HIV-Positive and HIV-Negative Presumptive Tuberculosis Patients. Am J Trop Med Hyg 2018;98(1):266-273. DOI: 10.4269/ajtmh.17-0486.
19. Fentress M, Henwood PC, Maharaj P, et al. High sensitivity of ultrasound for the diagnosis of tuberculosis in adults in South Africa: A proof-of-concept study. PLOS Glob Public Health 2022;2(10):e0000800. DOI: 10.1371/journal.pgph.0000800.
20. Belard S, Taccari F, Kumwenda T, Huson MAM, Wallrauch C, Heller T. Point-of-care ultrasound for tuberculosis and HIV—revisiting the focused assessment with sonography for HIV-associated tuberculosis (FASH) protocol and its differential diagnoses. Clinical Microbiology and Infection 2024;30(3):320-327. DOI: https://doi.org/10.1016/j.cmi.2023.10.021.
21. Ndege R, Weisser M, Elzi L, et al. Sonography to Rule Out Tuberculosis in Sub-Saharan Africa: A Prospective Observational Study. Open Forum Infect Dis 2019;6(4):ofz154. DOI: 10.1093/ofid/ofz154.
22. Bark CM, Dietze R, Okwera A, Quelapio MI, Thiel BA, Johnson JL. Clinical symptoms and microbiological outcomes in tuberculosis treatment trials. Tuberculosis (Edinb) 2011;91(6):601-4. DOI: 10.1016/j.tube.2011.05.007.
23. Mtafya B, Sabi I, John J, et al. Systematic assessment of clinical and bacteriological markers for tuberculosis reveals discordance and inaccuracy of symptom-based diagnosis for treatment response monitoring. Front Med (Lausanne) 2022;9:992451. DOI: 10.3389/fmed.2022.992451.
24. Loh LC, Lim BK, Wan Yusuf S. Significant resolution of tuberculous pleural effusion on chemotherapy alone. J R Coll Physicians Edinb 2010;40(2):100-4. DOI: 10.4997/JRCPE.2010.202.
25. Hunter L, Belard S, Janssen S, van Hoving DJ, Heller T. Miliary tuberculosis: sonographic pattern in chest ultrasound. Infection 2016;44(2):243-6. DOI: 10.1007/s15010-015-0865-8.
26. Safai Zadeh E, Görg C, Prosch H, et al. Lung Ultrasound and Pleural Artifacts: A Pictorial Review. Diagnostics 2024;14(2):179. (https://www.mdpi.com/2075-4418/14/2/179).
27. Allan-Blitz LT, Yarbrough C, Ndayizigiye M, Wade C, Goldsmith AJ, Duggan NM. Point-of-care ultrasound for diagnosing extrapulmonary TB. Int J Tuberc Lung Dis 2024;28(5):217-224. (In eng). DOI: 10.5588/ijtld.23.0471
Downloads
Additional Files
Published
Issue
Section
License
Copyright (c) 2026 Robert Ndege, Farida Bani, Omary Ngome, Mohamed Sasamalo, Dorcas Mnzava, Fiona Vanobberghen, Daniel H Paris, Maja Weisser, Martin Rohacek (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Authors retain the copyright for their published work. No formal permission will be required to reproduce parts (tables or illustrations) of published papers, provided the source is quoted appropriately and reproduction has no commercial intent.











